Wilms Tumour (Nephroblastoma) – Brief information
A Wilms tumour (Nephroblastoma) is a malignant tumour of the kidney. This text provides information about the characteristics of this disease, its frequencies, causes, symptoms, diagnosis, treatment, and prognosis.
Author: Maria Yiallouros, Editor: Maria Yiallouros, Reviewer: Prof. Dr. med. Norbert Graf, English Translation: Dr. med. Gesche Riabowol (nee Tallen), Last modification: 2024/06/05 https://dx.doi.org/10.1591/poh.wilms-patinfo.kurz.1.20120611
Table of contents
General information on the disease
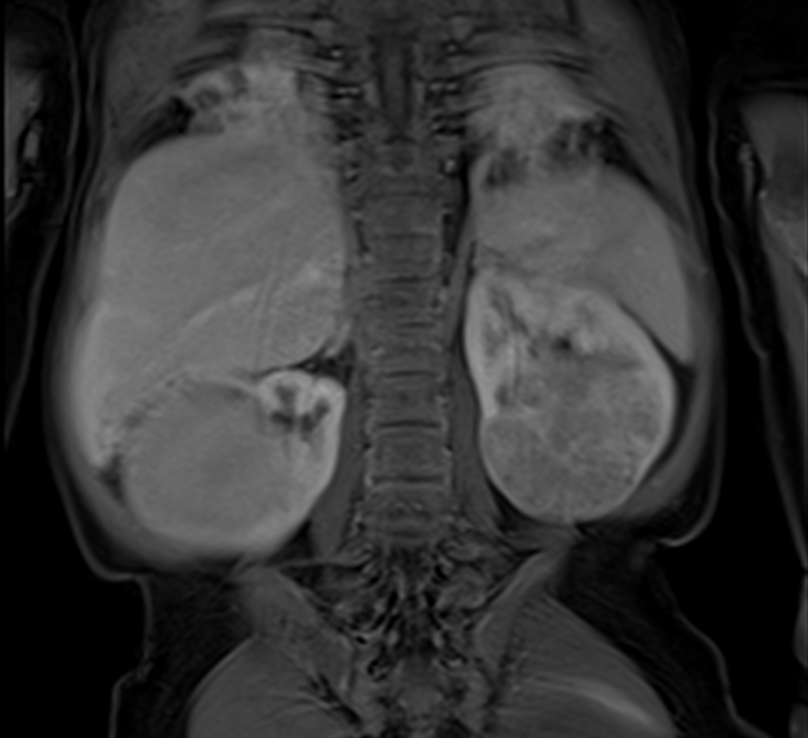
A Wilms tumour or nephroblastoma (nephro- for kidney, -blastoma for tumour), is a malignant solid tumour of the kidney. It is named after the German surgeon Dr. Max Wilms, who first described this tumour in the 19th century.
Wilms tumour arises from malignantly transformed primitive tissue. It resembles embryonal kidney tissue and is comprised of different tissue types. Usually, it mostly contains precursor kidney cells (blastema), but different developmental stages of various other tissue types, such as connective tissue, muscle, cartilage or epithelial tissue, to name a few, may also be present. This is why Wilms tumours are also called „mixed tumours“.
Wilms tumours are considered as malignant, because they grow fast and spread (metastasise) early. About 15 % of Wilms tumour patients (in Germany only 11% thanks to the scheduled routine check-ups) present with metastases [see metastasis] at tumour diagnosis. Most of these metastases can be found in the lymph nodes close to the kidney as well as in lung and liver. About 5 % of children with Wilms tumour have tumours in both kidneys (bilateral disease) at the time of diagnosis. These usually arise from so-called nephrogenic remainders, immature (embryonal) kidney tissue, which is known to be a precursor of Wilms tumour.
Wilms tumour is associated with other developmental disorders and/or with hereditary cancer syndromes more frequently than other cancers (see chapter „Causes“).
Incidence
Wilms tumour is the most common malignant kidney tumour of childhood. Depending on the age group (0-14 years or 0-17 years), it accounts for approximately 5.2 % or 4.2 %, respectively, of all malignant diseases in childhood and adolescence, thereby representing one of the relatively frequent solid tumours in these age group. According to the German Childhood Cancer Registry, about 95 children and teenagers under 18 years of age are newly diagnosed with a Wilms tumour each year. Hence, 8 or 9 out of 1,000,000 children and adolescents are diagnosed with the disease before they turn 18 years old.
As Wilms tumours are embryonal tumours, they occur mostly in young children: about 60 % of the patients are between 1 and 4 years old at diagnosis, 15 % are babies. The average age of the patients is 3 years, with girls being slightly more affected than boys. Nevertheless, older children and adolescents, adults rather seldomly, may also be affected.
Causes
The reasons why some children develop Wilms tumour, while others don’t, are not completely understood. It is well-known, however, that alterations of certain genes and chromosomes contribute to Wilms tumour development. Best-known is the so-called Wilms tumour gene 1 (WT1-gene). It plays a key role in normal kidney development. If it is mutated, thus dysfunctional, it can result in tumour development and other disorders. Additional candidate Wilms tumour genes have been described to be located on chromosome 11 and other chromosomes. Also, other chromosomal modifications have been associated with an increased risk of developing Wilms tumour. According to current knowledge, multiple genetic changes (mutations) need to occur before a Wilms tumour develops.
Children with certain inherited diseases (such as WAGR syndrome, Beckwith-Wiedemann syndrome (BWS) and hemihypertrophy, Denys-Drash syndrome (DDS), or Perlman syndrome) have a higher risk of developing a Wilms tumour than their healthy peers. Since these genetic conditions are not only characterized by various kinds of developmental abnormalities, but also associated with an elevated risk for tumour development, they are also known as "cancer predisposition syndromes". About 10-15 % of all Wilms tumours are associated with such cancer predisposing syndromes.
In addition, there are families in whom Wilms tumours, but no associated syndromes, are diagnosed frequently, due to hereditary predisposition. This condition affects about 1-2 % of patients, particularly those with bilateral disease. In the majority of children, however, the tumour develops newly (sporadically), meaning that it is not associated with any underlying cancer syndrome or hereditary predisposition. According to current knowledge, environmental factors do not play a role in Wilms tumour development.
Good to know: there are specific screening guidelines for patients who are genetically proned to potentially develop a Wilms tumour. Genetic counselling with a human geneticist is also necessary (see chapter „Diagnosis“).
Symptoms
Wilms tumours do not cause any health problems or pain for quite a while. Children typically present with a rounded tummy, which may sometimes be misdiagnosed as a sign of good nutrition. In about 10 % of children, a painless abdominal mass is randomly felt by the doctor during a child health check-up, while no other symptoms are being noticed. Less common symptoms indicative of Wilms tumour may include tummy pain, blood in the urine, fever, nausea, vomiting and indigestion, as well as weight loss, hypertension and persistent cough as a result of lung metastasis. Also, concomitant physical abnormalities or hereditary syndromes can be associated with Wilms tumour (see chapter „Causes“).
Diagnosis
If the doctor thinks that the young patient’s history (anamnesis) and physical examination are suspicious of a kidney tumour like the Wilms tumour, he will refer the child immediately to a hospital with a childhood cancer program (paediatric oncology unit), where further diagnostics can be initiated and performed by childhood cancer experts. Close collaboration between various specialists (such as paediatric oncologists, paediatric neurosurgeons, paediatric radiologists, to name a few) is required, both to find out whether the patient indeed suffers from a kidney tumour and, if so, to determine the tumour type and the extent of the disease. Knowing these details is absolutely essential for optimal treatment planning and assessment of prognosis.
Diagnostic imaging
Besides physical exam, diagnostic imaging procedures such as abdominal ultrasound, magnetic resonance imaging (MRI) and computed tomography (CT) play a major role in Wilms tumour diagnostics. They help determine tumour size and extent as well as differentiate (with a certainty of over 95 %) between a Wilms tumour and other, rather rare, kidney tumours (such as clear-cell sarcoma, rhabdoid tumour of the kidney, renal cell carcinoma and congenital mesoblastic nephroma, to name a few). Other cancers, such as lymphoma and neuroblastoma, can be found in the kidney area as well.
For obtaining the accurate diagnosis by imaging, however, high-quality equipment is required as well as an experienced radiologist to analyse the results. This is crucial because in Europe (in contrast to North America) tumour tissue is usually removed and examined after cessation of a pre-operative chemotherapy, meaning, histological confirmation of the diagnosis is done after therapy has already started (see chapter “Obtaining a tumour sample”).
Further tests to confirm diagnosis and assess tumour spread
Sometimes, diagnostic imaging alone is not sufficient to accurately differentiate Wilms from other tumours, such as neuroblastoma. For those patients, the doctors may recommend special additional tests. For example, in order to differentiate from neuroblastoma, a scintigraphy with the low radio-labelled substance 123-Iodo-meta-iodobenzylguanidine (123I-MIBG scintigraphy) may be indicated, or determining tumour markers characteristic for neuroblastoma, but not nephroblastoma, in a blood sample.
Other additional tests help determine whether the tumour has spread and, if so, where. For example, searching for lung metastases requires a chest X-ray or CT scan. In case of suspected liver, abdominal or brain metastases, magnetic resonance imaging is required.
Tests before treatment begins
Depending on the type of treatment being considered, further tests are needed in order to assess the condition of different organs. For example, prior to chemotherapy, the doctors will recommend an ultrasound of the heart (echocardiography), a hearing test (audiometry) as well as special diagnostics for determining kidney function (renal scintigraphy). Any changes occurring during the course of treatment can be assessed and managed better based on the results of those initial tests, which thus help to keep the risk of certain treatment-related side-effects as low as possible.
Obtaining a tumour sample (biopsy)
If it is possible to obtain accurate diagnosis by diagnostic imaging, the doctors will recommend to first proceed with chemotherapy (4 - 6 weeks) and obtain sample tumour tissue (biopsy) for microscopic and molecular genetic examinations later. The biopsy is then usually performed during surgical tumour removal. Only in a few situations, tumour tissue is required to be taken prior to chemotherapy (by means of punch biopsy or fine needle biopsy).
Good to know: Not all of the above-mentioned tests apply to every single patient. On the other hand, additional tests not mentioned here may be required individually. Ask the doctor which diagnostics are necessary and why.
Screening for patients with genetically predisposed tumour risk
Patients with certain hereditary predispositions for developing cancer (cancer predisposition syndromes) (see chapter “Causes”) can present with Wilms tumour and/or with nephroblastomatosis, a preliminary stage of Wilms tu-mour. They also have an increased risk of developing bilateral tumours (bilateral nephroblastoma). In addition, certain predisposing syndromes (such as Denys-Drash syndrome) are associated with kidney function impairments or even early kidney failure.
For these reasons, patients with predisposing syndromes or with kidney malformations associated with an increased risk of developing Wilms tumour are recommended to undergo quarterly clinical exam and diagnostic imaging (sonography). Due to the additional risk of bilateral disease in patients with these syndromes, unilateral and bilateral nephroblastoma are treated the same (see chapter “Course of treatment”). For all patients with a cancer predisposition syndrome or familial nephroblastoma, molecular diagnostics following genetic counselling are recommended.
Treatment planning
After the diagnosis has been confirmed, therapy is planned. In order to provide a highly individual, risk-adapted treatment regimen for the patient, the doctors consider certain individual factors that can influence the patient’s prognosis (risk factors or prognostic factors).
The most important prognostic factors for patients with a Wilms tumour are:
- the stage of the disease, that is, the extent of the tumour at diagnosis and the extent of its surgical removal;
- the type of Wilms tumour, that is, the characteristics of the tumour cells seen under the microscope, which describe, for example, how fast the tumour is expected to grow, thereby providing insight into its degree of malignancy.
Also, the response of the disease to preoperative therapy has major impact on prognosis. This response is partially dependent on stage and type of the disease. Scientists have also found out that, in addition to the microscopic characteristics of a Wilms tumour, its molecular genetics can also influence the course of the disease. Thus, certain gene and chromosome alterations in tumour cells appear to correlate with comparably more unfavourable outcomes.
When planning the treatment, the doctors consider all these individual factors in order to provide optimal therapy, thus best possible outcome for every patient.
Stages of Wilms tumour
The stage of the disease represents a major criterion for treatment planning. Aspects like tumour extent, involvement of one versus both kidneys, involvement of local lymph nodes and/or blood vessels, or even spread to distant organs are all included in the staging of a Wilms tumour.
The extent of surgical tumour removal is another essential staging criterion. Hence, accurate definition of the tumour stage is only possible after surgery. Prior to treatment, a preliminary classification, based on diagnostic imaging results at diagnosis, is done by differentiating between localized tumours in one kidney, tumours with metastases and/or bilateral tumours.
The staging system used in German treatment centres is according to the International Society of Pediatric Oncology (SIOP). It differentiates five stages of Wilms tumour (see table below).
|
Stage |
Definition |
|---|---|
|
Stage I |
The tumour is limited to the kidney. The outer layer of the kidney (renal capsule) did not break open. The tumour can be extracted completely. |
|
Stage II |
The tumour is not limited to the kidney, but has spread to blood vessels in the renal sinus or outside the area of the kidney. The tumor can be removed completely. Lymph nodes are not involved. |
|
Stage III |
The tumour cannot be removed completely or it has spilled or spread to local lymph nodes or it broke open (rupture of the tumour tissue). There is no spread to distant organs. |
|
Stage IV |
The tumour has spread through the blood (metastasised) to distant organs, such as the lungs, liver, bones and/or brain. |
|
Stage V |
Cancer cells are found in both kidneys (bilateral nephroblastoma). |
Notes to staging: a local stage (I-III) is also determined for stages IV and V, since this is decisive for choosing the appropriate post-surgical treatment. In case of bilateral tumours, the one with the highest local stage counts for the choice of post-surgical treatment.
Histological classification of Wilms tumour
Wilms tumours may look very different under the microscope (histologically), depending on which types of tissue they consist of and how mature (differentiated) the tumour cells are. The histology of the tumour determines the patient’s response to treatment and, hence, also affects prognosis. Therefore, it is crucial for appropriate treatment planning to determine the tumour’s histology, thus differentiating not only between Wilms tumour and any other type of kidney tumours, but also between different histological subtypes of Wilms tumour.
Based on how they appear under the microscope, Wilms tumours are classified into the following three histological groups reflecting the disease's degree of malignancy:
- nephroblastoma – low grade malignancy (favourable histology)
- nephroblastoma – intermediate grade of malignancy (standard histology)
- nephroblastoma – high grade of malignancy (unfavourable histology)
Certain subtypes of Wilms tumour are assigned to each of these groups. The exact way of assignment (classification) is based on whether tumour tissue is examined and assessed before or after chemotherapy. In Germany and other European countries, the removal and examination of tumour tissue usually (i.e. with the majority of patients) occurs after preoperative chemotherapy.
Treatment
Treatment of children and adolescents with Wilms tumour should take place in a children's hospital with a paediatric oncology program. Only such a childhood cancer centre provides highly experienced and qualified staff (doctors, nurses and many more), since they are specialised and focussed on the diagnostics and treatment of children and teenagers with cancer according to the most advanced treatment concepts. The doctors in these centres collaborate closely with each other. Together, they treat their patients according to treatment plans (protocols) that are continuously optimised.
The major goal of treatment is to eliminate the cancer while keeping the risk of side-effects and late sequelae as low as possible.
Treatment methods
Treatment of children and adolescents with Wilms tumour mainly consists of a combination of surgery and chemotherapy. Only a few patients receive additional radiotherapy.
Most patients with Wilms tumour receive chemotherapy first. The goal is to shrink the tumour to make it more easily removable by surgery. Some patients undergo surgery first. Surgery aims at complete removal of the tumour and possible metastases [see metastasis]. Usually, it is followed by another round of chemotherapy. Depending on the tumour staging after surgery and on possible spread, radiotherapy is required as well.
The individual treatment choice is based on the special features the tumour reveals under the microscope (subtype of Wilms tumour) as well as on the tumour extent after surgery (tumour stage). Taken together, the higher the degree of malignancy and the more advanced the tumour stage, the more intensive therapy will be. Since treatment of Wilms tumour can be associated with acute adverse effects, patients receive supportive therapy during cancer treatment in order to prevent and/or treat those side effects. Here you will find information on supportive care as well as recommendations for home, the latter of which may be helpful during or after chemo- and radiotherapy.
Course of treatment
The international and national (German) paediatric cancer societies (SIOP and GPOH) recommend the following treatment strategies:
Chemotherapy prior to surgery
In Germany as well as in several other European and non-European countries, treatment for all patients between six months and 16 years of age begins with chemotherapy (preoperative or neoadjuvant chemotherapy). The reason for this strategy is that chemotherapy usually helps to reduce tumour size, thereby making it easier for the surgeon to later remove the tumour. Also, the risk of tumour spill during surgery, thus spreading tumour tissue in the abdomen, is lower when the tumour is smaller. Also, radiotherapy after surgery may be waived.
In order to eliminate as many cancer cells as possible, patients receive a combination of agents that have proven to inhibit Wilms tumour growth (cytostatic agents). Some of these are vincristine and actinomycin D, to name a few. For some patients, for example children with metastases or highly malignant tumours, additional chemotherapy with an anthracycline (doxorubicine) is recommended. Preoperative chemotherapy usually takes four weeks, for patients with metastasised disease six weeks and for patients with bilaterial tumour up to a total of twelve weeks.
For patients with bilateral Wilms tumour, treatment duration is determined individually with the major goal to save both kidneys. Hence, intensification of the pre-operative chemotherapy using additional agents (such as etoposide and carboplatin) may be indicated. The same concept applies for patients with cancer predisposition syndromes: the goal is to salvage as much functional kidney tissue as possible.
Infants under the age of six months as well as teenagers older than 16 years do not receive preoperative chemotherapy. This is because most kidney tumours in these age-groups are not nephroblastoma, but other types of kidney tumours (such as renal cell carcinoma or congenital mesoblastic nephroma), which require a different type of treatment than Wilms tumour patients. Therefore, obtaining tumour tissue by surgery first is crucial for choosing the appropriate treatment strategy for these patients.
Surgery
The major aims of surgery are complete tumour removal as well as obtaining tissue samples for microscopic and molecular genetic analyses and assessing the extent of tumour growth.
The surgical approach is mainly dependent on whether only one or both kidneys are affected:
Patients with a Wilms tumour in only one kidney (unilateral tumour) usually undergo removal of the kidney with the tumour. Accordingly, this method is called tumour nephrectomy. As a result, the remaining kidney will enlarge over the following weeks and months, thereby becoming capable of fully taking over the removed one's function. Therefore, it is important to protect the remaining kidney from any damage (for example chronic inflammation) later in life. Kidney-preserving surgery can be sometimes achieved by an experienced surgeon, who is capable of completely removing the tumor out of the kidney. This applies mostly to stage I disease (see chapter „Therapy planning“).
If a patient presents with tumours in both kidneys (bilateral Wilms tumour) or has a high risk of developing a bilateral disease (for example, due to a hereditary cancer predisposition syndrome), the doctors will assess treatment options individually with the goal to at least save one of the affected organs.
For patients who developed lung metastases after chemotherapy, surgical removal of those is a feasible approach as long as it is performed by paediatric surgeons or urologists with expertise.
Chemotherapy after surgery
Following surgery, treatment usually carries on with chemotherapy for most patients (postoperative or adjuvant chemotherapy). Only for children with a tumour of low-grade malignancy that was limited to the kidney and could be completely removed (stage I and favourable histology), treatment is over after surgery. All other patients will receive a more or less intensive chemotherapy, depending on tumour type and weight at surgery as well as on local tumour stage.
For children with tumours of intermediate grade of malignancy that have not spread to other parts of the body (standard histology, stages I-III), therapy is usually continued by giving only two different cytostatics (vincristine and actinomycin D). If the disease has been classified as highly malignant and is at high stage, a combination of up to four different agents will be recommended (for example doxorubicine, carboplatin, etoposide and cyclophosphamide). The duration of therapy may be between one month (for patients with stage I tumours and standard histology) and eight to ten months (for patients with tumours of unfavourable histology or distant spread after surgery). For bilateral tumours, treatment may last up to one year in case of nephrogenic remainders (pre-stages of Wilms tumour) have been diagnosed by a pathologist.
Radiotherapy
Due to the development of efficient combination chemotherapy regimens as well as the introduction of preoperative (neoadjuvant) chemotherapy, most patients can do without radiotherapy (radiotherapy). For a few children, however, radiotherapy following surgery and chemotherapy is still recommended. These include patients with tumours of intermediate grade of malignancy (stage III) and of high grades of malignancy (stages II and up) (see chapter "Treatment planning).
Radiotherapy is usually carried out with doses between 15 and 30 gray (Gy) to the tumour site, depending on histological diagnosis (microscopical features) after surgery. Patients with residual tumours after surgery and chemotherapy usually receive an extra "boost" to the tumour site. For children with tumours that ruptured during surgery, radiotherapy of the whole abdomen is recommended with doses of 20 or 30 Gy, again depending on histology. Patients with tumour spread to the lungs usually receive chest irradiation as well. Patients with remaining lung metastases after pre-operative chemotherapy and surgery will also receive chest irradiation. In case of bilateral tumours, remaining kidney tissue and intermediate degree of malignancy, decreased radiation doses may be given (below 12 Gy) in order to maintain the remaining kidney function as long as possible.
Trials and registries for treatment optimisation
In the large paediatriac treatment centres, children and adolescents with Wilms tumour receive therapy according to standardised treatment protocols, which are designed and controlled by experts. They aim at continuously increasing survival rates while also reducing the risk of therapy-related late effects. Treatment according to such treatment protocols usually occurs within so-called therapy optimising trials.
In August 2011, a long-standing therapy optimising trial for children and adolescents with nephroblastoma ended: study SIOP WT 2001/GPOH. About 100 German, Austrian and Swiss treatment centres had participated in this study. It had been conducted under the roof of the German Society of Paediatric Oncology/Haematology (GPOH) by the study centre for nephroblastoma, as part of the International Nephroblastoma trial of SIOP (International Society of Pediatric Oncology).
In the meantime, based on the results of previous national and international trials, a new SIOP study for children and adolescents with kidney tumours has been developed (UMRRELLA SIOP-RTGS 2016). The trial has already been open for recruitment since 2019 in a few European countries, and in parts of Germany since late 2021. Until active in all Germany, patients with Wilms tumour treated in not yet participating centres will be registered with a registry and treated according to recommendations that have proven successful in the preceding trial. The German headquarters of trial and registry are located in the Children's Hospital of the University of Homburg/Saar, Germany. Principal Investigator is Prof. Dr. med. Norbert Graf.
For patients with metastasized disease, a randomized trial (RANDOMET 2017) has been open since beginning of 2022. The study analyses preoperative chemotherapy with the question whether a combination of vincristine, carboplatin and etoposide is as effective as the standard treatment with vincristine, actinomycin and doxorubicin. The goal is to reduce doxorubicin-induced cardiotoxicity in patients with metastatic disease, too, without jeopardizing cure rates.
Peculiarity of a clinical registry: Patients who cannot participate in any study, for example because none is available or open for them at that time, or since they do not meet the required inclusion criteria, respectively, may be included in a so-called registry. Such a registry pools scarce data in order to help with the planning of appropriate future clinical trials. To ensure optimal treatment for patients not registered in a study, experts from assigned trial panels usually provide recommendations and advice to the local caregiver team.
Prognosis
Long-term survival rates of children and teenagers after treatment of Wilms tumour are high. About 90 % of all Wilms tumour patients survive their disease today, thanks to modern diagnostic procedures and standardised combination therapies, which are continuously improved.
However, individual prognosis depends, above all, on the tumour subtype and the stage of disease. The lower the grade of malignancy of the tumour and the lesser advanced its stage, the more favourable the outcome. For example, the chances of survival for patients with a non-metastasised nephroblastoma of favourable or standard histology are currently higher than 90 %, while the outcomes of patients with highly malignant tumours are less favourable.
Nevertheless, under certain conditions, even patients with Wilms tumours of higher stages may have a good chance of survival. For example, prognosis for children whose disease has spread to other parts of the body (nephroblastoma stage IV) highly depends on how it responds to treatment: if preoperative chemotherapy and subsequent surgery result in complete elimination of the tumour and its metastases, survival rates of more than 80 % can be achieved.
Note: The above-mentioned numbers are statistical values. Therefore, they only provide information on the total cohort of patients with this type of tumour. They do not predict individual outcomes. Please ask the doctor, who is responsible for your child, for competent information on her individual prognosis.
Literature 
- Erdmann F, Kaatsch P, Grabow D, Spix C: German Childhood Cancer Registry - Annual Report 2019 (1980-2018). Institute of Medical Biostatistics, Epidemiology and Informatics (IMBEI) at the University Medical Center of the Johannes Gutenberg University Mainz 2020 [URI: https://www.kinderkrebsregister.de/ typo3temp/ secure_downloads/ 42507/ 0/ 1c5976c2ab8af5b6b388149df7182582a4cd6a39/ Buch_DKKR_Jahresbericht_2019_komplett.pdf]
- Furtwängler R, Graf N: Nierentumoren, in: Niemeyer C, Eggert A (Hrsg.): Pädiatrische Hämatologie und Onkologie. Springer-Verlag GmbH GDeutschland 2006, 2018 2. vollständig überarbeitete Auflage 2018, 441 [ISBN: 978-3-662-43685-1]
- Graf N: Nephroblastom (Wilms-Tumor). S1-Leitlinie 025/004 AWMF online 2016 [URI: https://www.awmf.org/ uploads/ tx_szleitlinien/ 025-004l_S1_Nephroblastom_Wilms-Tumor_2016-06-abgelaufen.pdf]
- Dome JS, Graf N, Geller JI, Fernandez CV, Mullen EA, Spreafico F, Van den Heuvel-Eibrink M, Pritchard-Jones K: Advances in Wilms Tumor Treatment and Biology: Progress Through International Collaboration. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2015 Sep 20; 33: 2999 [PMID: 26304882]
- Szavay P, Fuchs J, Leuschner I, Selle B, Graf N: Nierentumoren, in: Fuchs J (Hrsg.): Solide Tumoren im Kindesalter, Grundlagen – Diagnostik – Therapie. Schauttauer GmbH 2012, 111 [ISBN: 978-3-7945-2786-1]


 Brief information on Wilms tumour (PDF) (301KB)
Brief information on Wilms tumour (PDF) (301KB)