Osteosarcoma (Brief information)
Osteosarcoma are the most frequent bone tumours in childhood and adolescence. This text provides information about the characteristics of this disease, its frequencies, causes, symptoms, diagnosis, treatment, and prognosis.
Author: Maria Yiallouros, Editor: Maria Yiallouros, Reviewer: Prof. Dr. med. Stefan Bielack, Dr. med. Dorothee Carrle, Dr. Stefanie Hecker-Nolting, English Translation: Dr. med. Gesche Riabowol (nee Tallen), Last modification: 2025/11/19 https://dx.doi.org/10.1591/poh.patinfo.osteosarkom.kurz.20101215
Table of contents
General information on the disease
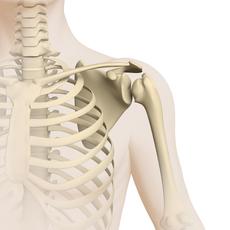
Osteosarcomas are rare malignant bone tumours. They are solid tumours arising from malignantly transformed bone-forming cells. Since osteosarcomas develop directly from bone tissue, they are also called primary bone tumours in order to distinguish them from cancers of other body parts that have spread to the bones (bone metastases). Most osteosarcomas grow and spread very quickly. Without an appropriate treatment, the disease is always fatal.
Incidence
Osteosarcomas are the most frequent malignant bone tumours in childhood and adolescence. According to the German Childhood Cancer Registry (Mainz, Germany), about 4 of 1,000,000 (in total approximately 60) children and adolescents under 18 years of age are diagnosed with osteosarcoma in Germany per year, which accounts for about 2.5 % of all paediatric malignancies in this age group.
Osteosarcoma can develop at any age. In childhood and adolescence (0–17 years), the frequency of this type of cancer peaks during the pubertal growth spurt in the second decade of life. The average age of patients within this age group is 14 years (when considering all patients, it is 16–18 years). Overall, boys are more frequently affected by the disease than girls (gender ratio: 1.2:1). Children under the age 5 are very rarely affected.
Localisation and spread
Most osteosarcomas arise in a long bone of a leg or an arm (for example in the thigh, shin or upper arm), usually in the areas that are close to the joints, the so-called metaphyses (metaphysis). In those bone areas (which include the so-called growth plates), longitudinal bone growth takes place, in particular during the pubertal growth spurt.
More than 50 % of all osteosarcomas are located around the knee (meaning above or below the knee joint). In general, osteosarcoma can develop in any bone. The tumour sometimes affects bone and/or bone marrow only, but it frequently also spreads (metastasizes) into adjacent tissue, such as connective, fat, muscle or nerve tissue. Rarely, an osteosarcoma does not arise from bone tissue, but from the surface of the bone (periosteal or parosteal) or even outside the bone (extraosseal or extraskeletal).
In about 10 to 20 % of children and adolescents with osteosarcoma, tumour spread (metastasis) is detectable at diagnosis. It has to be assumed for all other patients, too, that tumour cells have already spread elsewhere via the blood or lymphatic stream and, thus, have formed tiny metastases in other organs. These so-called micrometastases may not be detected at diagnosis due to their small size. Osteosarcomas primarily spread via the blood to the lungs (about 70 %), less frequently to bones and other organs. Sometimes, metastases develop both in lungs and bones at the same time. Metastasizing into the lymph nodes is extremely rare.
Very rarely (in less than 5 % of the patients), tumours are found in various different bones from the very beginning. This is called “multifocal disease”.
Histologic characteristics and tumour types
One major characteristic of osteosarcomas is that the tumour cells – in contrast to healthy bone-forming cells – produce immature bone tissue (osteoid). This feature alone allows to distinguish osteosarcoma from other bone tumours. Apart from that, however, the histologic characteristics of osteosarcoma (i.e. the way the tumour cells look under a microscope) are diverse as is the biological behaviour of these tumours. Most osteosarcomas in childhood and adolescence are highly malignant, since they grow rapidly and spread fast. Specialists also call them grade 3- (G3-) tumours. Only a few subtypes of osteosarcoma may be considered to be of low or medium grade of malignancy (so-called G1- or G2-tumours, respectively).
The World Health Organization (WHO) differentiates between the following subtypes of osteosarcoma based on their typical histologic features (WHO classification of bone tumours):
- conventional osteosarcoma (high grade of malignancy, G3)
- telangiectatic osteosarcoma (high grade of malignancy, G3)
- small-cell osteosarcoma (high grade of malignancy G3)
- low-grade central osteosarcoma (low grade of malignancy, G1)
- high-grade surface osteosarcoma (high grade of malignancy, G3)
- periosteal osteosarcoma (medium grade of malignancy, G2)
- parosteal osteosarcoma (usually low grade of malignancy, G1)
- secondary osteosarcoma (usually high grade of malignancy, G3)
Conventional osteosarcomas are the most frequent. They account for about 80 to 90 % of all osteosarcomas and are subclassified according to the current WHO classification (2020). All other subtypes are rather rare (accounting for less than 5 % each). The grade of malignancy has a major impact on treatment planning.
Extraosseal osteosarcomas are classified under the soft tissue sarcomas according to the WHO and are thus treated as those (see patient information on soft tissue sarcomas).
Causes
The causes of osteosarcoma are only partially understood. Genetic, epigenetic and growth-related factors are currently being considered to all contribute to the risk of developing osteosarcoma. Frequently, the tumour cells contain DNA changes, such as in so-called tumour suppressor genes (like the retinoblastoma gene or the TP53 gene), which normally prevent tumour formation. In general, the genetic defects in osteosarcoma cells are characteristically quite complex, being associated with impaired cell cycle control, cell communication or differentiation of bone-forming cells, respectively. Since osteosarcoma preferably develops during puberty, it is assumed that the growth signals that are associated with puberty may play a role in the development and progression of osteosarcoma.
Apart from that, further factors are known to elevate the risk of osteosarcoma. These include ionising radiation as used, for example, in radiation therapy, as well as certain cellular poisons (cytotoxins), such as certain anticancer agents that are applied during chemotherapy (cytostatics, especially so-called alkylants). Radiation and cytotoxins are capable of causing genetic instability in bone-forming cells, thereby contributing to initiate osteosarcoma development.
Some osteosarcoma can also be associated with so-called cancer predisposition syndromes, very rare hereditary diseases chararacterized by mutations that (compared to healthy individuals) are connected with a higher risk of developing a malignancy at a younger age. Cancer predisposition that play a role in the development of osteosarcoma are, for example, the hereditary (mostly bilateral) type of retinoblastoma as well as Li-Fraumeni syndrome or Bloom syndrome. Also, children with chronic bone diseases, such as Paget`s disease, have an increased risik to develop osteosarcoma.
For most patients, however, no particular cause can be found.
Symptoms
The most frequent symptoms of osteosarcoma are pain in a bone or joint and/or a (progressive) swelling over a bone or bony part of the body. Occasionally, the pain is not directly experienced at the tumour site, but rather in a different area of the affected skeletal part.
Pain may be intermittant and activity-dependen, but frequently also occurs at rest. About a quarter of the patients complain about pain at night. With increasing tumour growth, the pain may be accompanied by a visible and / or palpable lump at the tumour site (frequently in the area of the knee joint ). The swelling may be reddened and warm and may sometimes lead to impaired mobility, which initially may be mistaken for the result of a sports injury or bone inflammation. Sometimes, even a minor accident may cause a fracture at this site (pathological fracture). In about 5 to 10 % of patients, bone fracture is the first symptom of the disease.
The symptoms described above (such as pain, swelling, reddening) are caused by the tumour growth within the tender bone, which is very sensitive to pain, and adjacent soft tissue. Patients with advanced disease may also complain of general symptoms such as fever, weight loss and/or fatigue. For some patients, only a few weeks, for others up to several months may pass between first symptoms and the diagnosis of osteosarcoma.
Good to know: Not all children and adolescents presenting with the complaints described above suffer from osteosarcoma or any other malignant bone tumour. However, every type of musculoskeletal pain in a child or a teenager should be taken seriously and be dealt with by an experienced paediatrician in order to appropriately rule out an underlying cancer.
Diagnosis
If the paediatrician thinks that the young patient’s history (anamnesis) and physical examination are suspicious of a malignant bone tumour, the child should immediately be referred to a hospital with a childhood cancer program (paediatric oncology unit), where further diagnostics can be initiated and performed by childhood cancer professionals. Very close collaboration between various specialists (such as paediatric oncologists, paediatric surgeons, paediatric radiologists, to name a few) is required both to find out whether the patient really suffers from a malignant bone tumour and, if so, to determine the tumour type and the extension of the disease. Knowing these details is absolutely essential for optimal treatment planning and prognosis.
Clinical exams and laboratory tests
The caregiver team at the hospital will first take a thorough history followed by a physical exam, during which the location, size, consistency and range of motion of the affected site is being assessed. Lab tests rather play a secondary role in the diagnostics of osteosarcoma. However, serum levels of certain parameters may, if elevated, be indicative of an altered bone metabolism and, thus, a potential bone tumour. These include, for example, the alkaline phosphatase (AP) and the lactate dehydrogenase (LDH). Since elevated AP and LDH levels can be associated with different (bone) diseases, they only add to other diagnostic tests and are not specific for bone tumours.
Imaging tests for tumour detection
Based on their typical radiological features, most malignant bone tumours can already be diagnosed on an X-ray examination Additional imaging procedures such as magnetic resonance imaging (MRI) subsequently help to define the exact tumour site and size as well as its demarcation with regard to adjacent tissue (such as blood vessels, muscles, nerves, tendons or joints). Nearby metastases (called skip metastases) are easily detectable by these methods, too. For imaging of affected soft tissue and bone marrow, MRI is superior to computed tomography (CT). Apart from plain X-rays, it is therefore considered as the gold standard in radiological diagnosis of osteosarcoma.
Tissue removal and analysis
For final confirmation of the diagnosis, tumour tissue is required to be removed (biopsy). The biopsy should only be performed by doctors who are specialized in surgery of bone tumours. This ensures that the access chosen for biopsy does not cause problems for subsequent treatment.
An unfavourably planned biopsy may result in the requirement of a far more extensive subsequent surgery than initially necessary or, worst case scenario, in the inoperability of a tumour that would have been initially removable (thereby requiring limb amputation, for example). Also, suboptimally performed biopsy may be associated with an increased risk of recurrent disease. Hence, it is recommended that, even prior to biopsy, all patients with a suspected bone tumour are referred to a bone tumour reference centre or a centre that belongs to an osteosarcoma network.
In order to obtain a sufficient amount of tumour tissue, open surgery, meaning the removal of tissue during the surgical procedure for tumour resection, is gold-standard. Needle biopsy may be considered for in single cases only. In these situations, special needles are used (tru-cut biopsy). The obtained samples are subsequently analysed both histologically and immunohistochemically by specialists. These analyses serve to confirm the diagnosis of osteosarcoma and, once confirmed, to determine the subtype.
Tests to assess tumour spread
Once the diagnosis of osteosarcoma has been confirmed, further imaging tests are required to find out if and to which extent the cancer has spread and which organs are involved. Since most metastases develop in the lungs, a chest x-ray as well as a chest-CT are indispensable. In order to assess or, respectively, rule out bone metastases, a skeletal scintigraphy (bone scan) using radioactive technetium (11mTc) is performed. In addition (or instead), positron emission tomography (PET) with radioactively labelled glucose (18F-fluoride-deoxyglucose, short FDG) may be an option. PET is either combined with computed tomography (PET-CT) or magnetic resonance imaging (PET-MRI).
Tests before treatment begins
Before treatment begins, further tests are needed in order to assess the condition of different organs. Therefore, the doctors will recommend an electrocardiography (ECG) as well as an ultrasound of the heart (echocardiography), a hearing test (audiometry), special diagnostics for determining kidney and lung functions as well as various blood tests. Any changes occurring during the course of treatment can be assessed and managed better based on the results of those initial tests, which thus help to keep the risk of certain treatment-related side effects as low as possible.
Good to know: Not all of the above-mentioned tests will be done for every single patient. On the other hand, additional tests not mentioned here may be required individually. Ask the doctor which diagnostics are necessary and why.
Treatment planning
After the diagnosis has been confirmed, therapy is planned. In order to design a highly individual, risk-adapted treatment regimen for the patient, certain individual factors influencing the patient’s chance of recovery (prognosis) – called risk factors or prognostic factors – are being considered during treatment planning (risk-adapted treatment strategy).
Important prognostic factors are the type, the localisation, size and spread of the tumour, which are assessed before treatment starts. In the course of treatment, additional prognostic factors, such as the extent of tumour removal (incomplete versus complete) and the response of the disease to chemotherapy, are of major impact on how treatment is going to be continued. Taking into account all these factors aims at achieving the best outcome possible for each patient.
Treatment
Treatment of children and adolescents with osteosarcoma should take place in a children's hospital with a paediatric oncology program that is also part of a specialized osteosarcoma-network. Only such a childhood cancer centre provides highly experienced and qualified staff (doctors, nurses and many more), since they are specialised and focussed on the diagnostics and treatment of children and teenagers with cancer according to the most advanced treatment concepts. The doctors in these centres collaborate closely with each other. Together, they treat their patients according to treatment plans (protocols) that are continuously optimised. The goal of the treatment is to achieve good cure rates while avoiding side effects as much as possible.
Treatment methods
Treatment of children and adolescents with osteosarcoma basically consists of surgery (local therapy) and chemotherapy. Only patients with the rare low-grade subtypes may benefit from surgery alone under certain conditions. Radiotherapy plays an inferior role. It is only considered when complete tumour removal is not possible. Overall, treatment takes about nine to twelve months.
Goal of surgery is to remove the tumour. Chemotherapy uses drugs (so-called cytostatic agents or cytostatics) that can kill fast-dividing cells, such as cancer cells, or inhibit their growth, respectively. High-dose radiation therapy is only required if gross total tumour resection is not an option and is done using energy-rich, electromagnetic radiation, given through the skin to the tumour region. Radiation causes DNA damage in tumour cells, thereby leading to cell death.
In order to prevent or adequately manage the side effects of the intensive therapy, specific supportive care regimens (see (supportive therapy) have been established. Here you will find information on supportive care as well as recommendations for home, the latter of which may be helpful during or after chemo- and radiotherapy.
Course of treatment
The following treatment plan is currently the international standard for patients with highly malignant osteosarcoma: The first step includes a preoperative (neoadjuvant) chemotherapy for a total of about two to three months. The subsequent surgery for tumour removal is followed by another (adjuvant) chemotherapy,which lasts about six months (individual treatment plans may apply to single patients). Requirement of radiotherapy is the exception and needs individually adjusted decision-making. Removal of lesions sus-picious of metastasis is usually done after surgery of the primary tumour. The different treatment phases will be outlined in the next paragraphs.
Chemotherapy prior to surgery
For most patients, therapy is initiated by a ten-week phase of chemotherapy (called preoperative, neoadjuvant or induction chemotherapy). The goal of this chemotherapy is to shrink the tumour and potential spread (metastases) in order to optimise the conditions for subsequent surgical tumour removal, thereby contributing to safety and efficacy of surgery. Also, chemotherapy helps eliminate micrometastases and prevent further tumour spread. This time is also used to prepare for surgery.
In order to eliminate as many tumour cells as possible, patients receive a combination of different chemotherapeutic agents (cytostatics) that have proven to be effective in the treatment of osteosarcoma. These agents include methotrexate (high dose, HD-MTX), adriamycin (ADR = doxorubicin) and cisplatin (DDP), abbreviated “MAP”. Chemotherapy is given as several courses over several days each (altogether two cycles of cisplatin/doxorubicin and four cycles of methotrexate), during which the patient needs to be inpatient. Between courses, patients can go home as outpatients. Only in case of severe side-effects, readmission is required.
Local therapy
Surgery is the gold standard for local therapy. (Additional) radiotherapy is an option only for tumours that cannot be completely removed due to their location or extent, respectively.
Surgery
Following chemotherapy and the shortest possible recovery period, patients undergo surgery with the goal of complete surgical tumour removal. Surgery aims at removing both tumour parts that have been identified by diagnostic imaging (macroscopic) as well as microscopically small ones, because gross total tumour resection is crucial for subsequent chances of cure. The term “gross total resection” is defined as the removal of all tumour parts including the biopsy scar, biopsy channel and a layer of surrounding healthy tissue.
Choice of the surgical technique depends on the site and extent of the tumour as well as the on response to chemotherapy and is adjusted to every patient individually. Also, metastases need to be removed to increase the chances of cure.
Thanks to major technical progress regarding limb-saving surgical procedures, tumour removal does usually not require amputation any more. Different techniques are available for limb reconstruction based on the location and the size of the tumour. For example, metal joint implants are frequently used; for children, also growing endoprotheses are available.
Following surgery, a pathologist examines the tumour tissue to find out how the disease has responded to the preceding chemotherapy. This response is assessed by measuring the amount of the tumour cells that are still alive. An amount of less than 10 % is considered as good response, which is achieved for about 50 % of all osteosarcoma patients. This response represents a very important prognostic factor. However, changing the chemotherapy regimen in case of poor response is not required.
Radiotherapy
For patients whose tumour and/or metastases [see metastasis] cannot be completely removed, radiation therapy might in some situations be an alternative or additional treatment option. However, radiotherapy is far less favourable than gross total tumour resection, and hence, all other options have to be carefully considered before. In general, osteosarcomas are not very sensitive to radiation, thereby requiring high radiation doses for treatment. Also, delivery of radiotherapy might me challenging depending on the tumour location.
The pros and cons of radiotherapy need interdisciplinary discussion and decision-making on an individual basis. In suitable cases, modern radiation techniques such as intensity modulated radiotherapy (IMRT), proton or heavy ion therapy may be used. The feasibility of heavy ion radiotherapy for children and adolescents is currently being investigated in the framework of a clinical study.
Chemotherapy after surgery
After surgery, chemotherapy with methotrexate, adriamycin and cisplatin (MAP) is continued for 18 weeks (postoperative chemotherapy). Treatment consists of 12 cycles (a total of two cycles cisplatin/doxorubicin, two cycles doxorubicin and eight cycles methotrexate) and takes about six to seven months including the treatment breaks.
Treatment of recurrent disease
About 20 to 40 % of patients with osteosarcoma develop recurrent disease. Like the primary disease, recurrent osteosarcoma (relapse) requires complete removal of the tumour in order to achieve cure. Subsequent treatment depends on the tumour sites and the timepoint of relapse (early versus late relapse).
Patients with single lung metastases, in particular if these occur later than two to three years after primary diagnosis, may benefit from surgery alone. Other patients with relapsed osteosarcoma will also need another round of chemotherapy, for example with carboplatin, etoposide and ifosfamide. In palliative settings, radiation therapy may be an option. Several clinical trials (phase I/II) are also investigating new agents (such as tyrosine kinase inhibitors). Overall, the prognosis for patients with relapsed osteosarcoma is not favourable.
Therapy optimising trials / registries
In the large paediatriac treatment centres, children and adolescents with osteosarcoma receive therapy according to standardised treatment plans (protocols). These protocols are designed by experts and aim at steadily improving the patients’ survival rates while also reducing the risk of therapy-related late effects. Therapy according to such treatment protocols is usually carried out within therapy optimising trials or registries.
Therapy optimising trials are standardised and controlled clinical trials that aim at steadily developing and improving treatment concepts for sick patients based on the current scientific knowledge. Patients who cannot participate in any study, for example because none is available or open for them at that time or since they do not meet the required inclusion criteria, respectively, are often included in a so-called registry. One goal of a registry is to collect treatment data for research questions. Furthermore, the registry centre supports the doctors at site with (non-commital) treatment recommendations based on the most recent data on best treatment options, in order to provide the patient with optimal therapy even without the framework of a clinical study.
In Germany, patients with osteosarcoma had the option to be enrolled in the therapy optimising trial EURAMOS 1 until June 2011. The study was conducted by the Cooperative OsteoSarcoma Study Group (COSS) of the German Society for Paediatric Oncology and Haematology (GPOH) in collaboration with other renowned groups. Numerous children’s hospitals and treatment centres all over Germany and other European as well as North American countries participated.
Since the EURAMOS trial has been closed for patient admission, children and adolescents with osteosarcoma are being registered in the so-called COSS Registry. The German headquarters of trial and registry are located at the Clinic for Paediatric Oncology, Haematology and Immunology of the Klinikum Stuttgart – Olgahospital, Germany. Since November 2023, the principal investigator has been Dr. med. Stefanie Hecker-Nolting.
The registry does not provide protocols for diagnostics and therapy. However, the study centres recommend the current standard treatment (MAP chemotherapy prior and after surgery, see chapter “Treatment”) for all patients who are not participating in a trial. These recommendations also include the diagnostic standards, supportive treatment and long-term care. Having patients participate in open clinical trials is endorsed. Your caregiver team will inform you about the current options.
Prognosis
The chances of survival (prognosis) for children and teenagers with an osteosarcoma depend on various factors. In particular, the subtype and the site of the tumour, its extent at diagnosis, response to preoperative chemotherapy as well as the extent of surgical tumour/metastasis removal are of major prognostic importance.
During the last five decades, prognosis of children and teenagers with osteosarcoma has significantly been increased by major improvements of diagnostics and treatment in the framework of therapy optimising trials.
By combining different treatment methods and particularly by introducing intensive, standardised combination chemotherapy regimens, survival rates of approximately 70 % can be achieved today. Favourable prognosis usually requires complete tumour removal and good response to the chemotherapy that precedes surgery.
Patients with non-metastasised osteosarcoma of the arm or leg have the most favourable prognosis – cure rates may reach more than 70 %, depending on the response to chemotherapy. Patients with good response (that means less than 10 % of living tumour cells after chemotherapy) have a much better prognosis than patients with poor response. The latter have a high risk of recurrent disease (relapse); the probability of disease-free survival is currently less than 50 %.
Patients with tumours of the trunk, which often cannot be completely removed because of their location, have a less favourable prognosis than patients with a tumour of the extremities (which generally can be removed more easily). The situation is similar for patients with large tumours that cannot be removed in total. In case of metastases, their number, localisation and operability are major prognostic factors. Patients with single, removable lung metastases have a better chance of survival than patients with bone metastases or multifocal disease. For patients with recurrent disease, prognosis is usually rather unfavourable (with a chance of survival of 20-25 %). However, cure of (even repetitive) recurrence is possible, as long as the tumours can be removed completely.
Note: The survival rates mentioned in the text above are statistical values. Therefore, they only provide information on the total cohort of patients with osteosarcoma. They do not predict individual outcomes. In the context of cancer, the term "cure" should rather be referred to as "free of cancer", because current treatment regimens may help to destroy the tumour, but they are also frequently associated with numerous late-effects. Early detection and appropriate management of these long-term secondary effects typically require intensive rehabilitation and thorough long-term follow-up care, although a patient may have been „cured“ from the cancer.
References 
- Ronckers CM, Spix C, Grabow D, Erdmann F: German Childhood Cancer Registry - Annual Report 2022 (1980-2021). Institute of Medical Biostatistics, Epidemiology and Informatics (IMBEI) at the University Medical Center of the Johannes Gutenberg University Mainz 2025 [URI: https://www.kinderkrebsregister.de/ fileadmin/ kliniken/ dkkr/ pdf/ jb/ jb2022/ JB_2022_final.pdf]
- Stanulla M, Erdmann F, Kratz CP: Risikofaktoren für Krebserkrankungen im Kindes- und Jugendalter. Monatsschrift Kinderheilkunde 169, 30-38 2021 [DOI: 10.1007/s00112-020-01083-8]
- Hecker-Nolting S: Osteosarkome. Leitlinie der Gesellschaft für Pädiatrische Onkologie und Hämatologie 2021 [URI: https://www.awmf.org/ uploads/ tx_szleitlinien/ 025-005l_S1_Osteosarkome_2021-11.pdf]
- WHO Classification of Tumours Editorial Board: Soft Tissue and Bone Tumours. 5th ed. Lyon: IARC Press 2020 [URI: https://publications.iarc.who.int/ Book-And-Report-Series/ Who-Classification-Of-Tumours/ Soft-Tissue-And-Bone-Tumours-2020]
- Smeland S, Bielack SS, Whelan J, Bernstein M, Hogendoorn P, Krailo MD, Gorlick R, Janeway KA, Ingleby FC, Anninga J, Antal I, Arndt C, Brown KLB, Butterfass-Bahloul T, Calaminus G, Capra M, Dhooge C, Eriksson M, Flanagan AM, Friedel G, Gebhardt MC, Gelderblom H, Goldsby R, Grier HE, Grimer R, Hawkins DS, Hecker-Nolting S, Sundby Hall K, Isakoff MS, Jovic G, Kühne T, Kager L, von Kalle T, Kabickova E, Lang S, Lau CC, Leavey PJ, Lessnick SL, Mascarenhas L, Mayer-Steinacker R, Meyers PA, Nagarajan R, Randall RL, Reichardt P, Renard M, Rechnitzer C, Schwartz CL, Strauss S, Teot L, Timmermann B, Sydes MR, Marina N: Survival and prognosis with osteosarcoma: outcomes in more than 2000 patients in the EURAMOS-1 (European and American Osteosarcoma Study) cohort. European journal of cancer 2019, 109: 36 [PMID: 30685685]
- Kager L, Bielack S: Osteosarkome. in: Niemeyer C, Eggert A (Hrsg.): Pädiatrische Hämatologie und Onkologie, Springer-Verlag GmbH Deutschland 2. vollständig überarbeitete Auflage 2018, 509 [ISBN: 978-3-662-43685-1]
- Casali PG, Bielack S, Abecassis N, Aro HT, Bauer S, Biagini R, Bonvalot S, Boukovinas I, Bovee JVMG, Brennan B, Brodowicz T, Broto JM, Brugières L, Buonadonna A, De Álava E, Dei Tos AP, Del Muro XG, Dileo P, Dhooge C, Eriksson M, Fagioli F, Fedenko A, Ferraresi V, Ferrari A, Ferrari S, Frezza AM, Gaspar N, Gasperoni S, Gelderblom H, Gil T, Grignani G, Gronchi A, Haas RL, Hassan B, Hecker-Nolting S, Hohenberger P, Issels R, Joensuu H, Jones RL, Judson I, Jutte P, Kaal S, Kager L, Kasper B, Kopeckova K, Krákorová DA, Ladenstein R, Le Cesne A, Lugowska I, Merimsky O, Montemurro M, Morland B, Pantaleo MA, Piana R, Picci P, Piperno-Neumann S, Pousa AL, Reichardt P, Robinson MH, Rutkowski P, Safwat AA, Schöffski P, Sleijfer S, Stacchiotti S, Strauss SJ, Sundby Hall K, Unk M, Van Coevorden F, van der Graaf WTA, Whelan J, Wardelmann E, Zaikova O, Blay JY, ESMO Guidelines Committee, PaedCan and ERN EURACAN: Bone sarcomas: ESMO-PaedCan-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of oncology : official journal of the European Society for Medical Oncology 2018 Oct 1; 29:iv79-iv95 [PMID: 30285218]
- Kager L, Tamamyan G, Bielack S: Novel insights and therapeutic interventions for pediatric osteosarcoma. Future oncology (London, England) 2017, 13: 357 [PMID: 27651036]
- Bielack SS, Hecker-Nolting S, Blattmann C, Kager L: Advances in the management of osteosarcoma. F1000Research 2016 Nov 25; 5: 2767 [PMID: 27990273]
- Marina N, Smeland S, Bielack S, Bernstein M, Jovic G, Krailo M, Hook J, Arndt C, van den Berg H, Brennan B, Brichard B, Brown K, Butterfass-Bahloul T, Calaminus G, Daldrup-Link H, Erksson M, Gebhardt M, Gelderblom H, Gerss J, Goldsby R, Goorin A, Gorlick R, Grier H, Hale J, Hall K, Hardes J, Hawkins D, Helmke K, Hogendoorn P, Isakoff M, Janeway K, Jürgens H, Kager L, Kühne T, Lau C, Leavey P, Lessnick S, Mascarenhas L, Meyers P, Mottl H, Nathrath M, Papai Z, Randall R, Reichardt P, Renard M, Safwat A, Schwartz C, Stevens M, Strauss S, Teot L, Werner M, Sydes M, Whelan J: Comparison of MAPIE versus MAP in patients with a poor response to preoperative chemotherapy for newly diagnosed high-grade osteosarcoma (EURAMOS-1): an open-label, international, randomised controlled trial . Lancet Oncol 2016, 17: 1396 [PMID: 27569442]
- Isakoff MS, Bielack SS, Meltzer P, Gorlick R: Osteosarcoma: Current Treatment and a Collaborative Pathway to Success. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2015 Sep 20; 33: 3029 [PMID: 26304877]
- Bielack SS, Smeland S, Whelan JS, Marina N, Jovic G, Hook JM, Krailo MD, Gebhardt M, Pápai Z, Meyer J, Nadel H, Randall RL, Deffenbaugh C, Nagarajan R, Brennan B, Letson GD, Teot LA, Goorin A, Baumhoer D, Kager L, Werner M, Lau CC, Sundby Hall K, Gelderblom H, Meyers P, Gorlick R, Windhager R, Helmke K, Eriksson M, Hoogerbrugge PM, Schomberg P, Tunn PU, Kühne T, Jürgens H, van den Berg H, Böhling T, Picton S, Renard M, Reichardt P, Gerss J, Butterfass-Bahloul T, Morris C, Hogendoorn PC, Seddon B, Calaminus G, Michelagnoli M, Dhooge C, Sydes MR, Bernstein M, EURAMOS-1 investigators: Methotrexate, Doxorubicin, and Cisplatin (MAP) Plus Maintenance Pegylated Interferon Alfa-2b Versus MAP Alone in Patients With Resectable High-Grade Osteosarcoma and Good Histologic Response to Preoperative MAP: First Results of the EURAMOS-1 Good Response Randomized Controlled Trial. Journal of clinical oncology 2015, 33: 2279 [PMID: 26033801]
- Whelan JS, Bielack SS, Marina N, Smeland S, Jovic G, Hook JM, Krailo M, Anninga J, Butterfass-Bahloul T, Böhling T, Calaminus G, Capra M, Deffenbaugh C, Dhooge C, Eriksson M, Flanagan AM, Gelderblom H, Goorin A, Gorlick R, Gosheger G, Grimer RJ, Hall KS, Helmke K, Hogendoorn PC, Jundt G, Kager L, Kuehne T, Lau CC, Letson GD, Meyer J, Meyers PA, Morris C, Mottl H, Nadel H, Nagarajan R, Randall RL, Schomberg P, Schwarz R, Teot LA, Sydes MR, Bernstein M, EURAMOS collaborators: EURAMOS-1, an international randomised study for osteosarcoma: results from pre-randomisation treatment. Annals of oncology : official journal of the European Society for Medical Oncology / ESMO 2015, 26: 407 [PMID: 25421877]
- Kager L, Bielack S: [Chemotherapeutic concepts for bone sarcomas]. Der Unfallchirurg 2014, 117: 517 [PMID: 24903502]
- Henrichs M-P, Hardes J, Jürgens H: Maligne Knochentumoren. in: Solide Tumoren im Kindesalter. Fuchs J (Hrsg.), Schattauer GmbH Stuttgart 2012, 330 [ISBN: 978-3-7945-2781-1]
- Bielack S, Carrle D: Diagnostik und multimodales Therapiekonzept des Osteosarkoms. ärztliches journal reise & medizin onkologie, Otto Hoffmanns Verlag GmbH 3/2007, S: 34
- Bielack S, Machatschek J, Flege S, Jürgens H: Delaying surgery with chemotherapy for osteosarcoma of the extremities. Expert Opin Pharmacother 2004, 5: 1243 [PMID: 15163270]
- Bielack S, Kempf-Bielack B, Delling G, Exner G, Flege S, Helmke K, Kotz R, Salzer-Kuntschik M, Werner M, Winkelmann W, Zoubek A, Jürgens H, Winkler K: Prognostic factors in high-grade osteosarcoma of the extremities or trunk. J Clin Oncol 2002, 20: 776 [PMID: 11821461]


 Brief information on Osteosarcoma (PDF) (330KB)
Brief information on Osteosarcoma (PDF) (330KB)