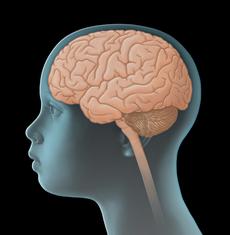
Ependymoma – Brief information
Ependymoma is a rare tumour of the central nervous system (CNS). This text provides information about the characteristics of this disease, its frequency, causes, symptoms, diagnosis, treatment, and prognosis.
Author: Dr. med. Gesche Riabowol (née Tallen) / Maria Yiallouros, Editor: Maria Yiallouros, Reviewer: Dr. med. Martin Mynarek, English Translation: Dr. med. Gesche Riabowol (nee Tallen), Last modification: 2026/02/04 https://dx.doi.org/10.1591/poh.patinfo.ependy.kurz.1.20070626
Table of contents
General information on the disease
Ependymomas are tumours of the central nervous system (CNS). They are solid tumours arising from malignantly transformed cells of the brain or spinal cord. Since they develop directly from CNS cells, they are also called primary CNS tumours in order to distinguish them from cancers of other body parts that have spread to the CNS (metastasis).
There are different types of ependymomas – some of which grow rather slowly, while others expand fast. Nevertheless, since for a growing mass the room in the bony skull is limited, even slow-growing ependymomas can become life threatening.
Localisation and spread in the central nervous system
Ependymomas arise from malignantly transformed cells of the ependyma. Ependyma cells are the cells that form the inner coat of the brain’s cavities (ventricles) as well as of the spinal canal. Therefore, most ependymomas are situated in the cerebral ventricles or the spinal canal.
The majority of ependymomas (about 60 %) are found in the fourth ventricle, which is located in the lower back of the skull (posterior cranial fossa). From there, they tend to grow toward the cerebellum, the brainstem and the cervical spine. The doctors call this infratentorial growth. About 30 % of ependymomas grow in the area of the so-called lateral ventricles in the cerebrum (supratentorial growth); 10 % are located in the spinal cord (intraspinal).
In less than 5 % of children with either supra- or infratentorial ependymoma, the tumour has already spread within the central nervous system (CNS) at the time of first diagnosis. Tumour spread (metastasis) outside the CNS, for example into the lung and/or lymph nodes, is rare.
It is well-known by now that ependymomas that grow in different parts of the central nervous system differ regarding their biological characteristics and the health problems they cause in a patient. This particularly applies to spinal ependymomas (see chapter „Treatment planning“).
Incidence
Accounting for barely 2 % of all malignancies in children and adolescents, ependymomas are overall rare. They comprise about 7 % of all primary central nervous system tumours in childhood and adolescence. In Germany, about 40 children and adolescents under the age of 18 years are newly diagnosed with ependymoma each year. This corresponds to an incidence rate of about 3 per 1,000,000 children / adolescents.
Although mostly affecting children in the first three to four years of life, ependymomas can generally occur in any age group. The patients’ average age at diagnosis is about five years. Boys are a slightly more affected than girls (gender ratio: 1,4 : 1). Spinal ependymomas are an exception, since they affect mostly adolescents: the mean age at diagnosis is 14 years.
Causes
The causes for the development of ependymoma are still unknown. In general, individuals who received radiotherapy of the brain when they were young, for example children with acute leukaemia or retinoblastoma, have an increased risk of developing a brain tumour later.
Ependymomas can also be associated with so-called cancer predisposition syndromes, hereditary diseases characterized by mutations that, compared to healthy individuals, are associated with a higher risk of developing a malignancy at a younger age. An example for such a hereditary cancer predisposition is neurofibromatosis type II (NF-2): children with this inherited disease have a higher risk of ependymoma in the spinal canal than their healthy peers. In addition, it has been shown that ependymoma are frequently associated with certain aberrations of genes or chromosomes within cells. The resulting impairments of cell development and cell communication may be contributing factors promoting the transformation of a healthy into a cancer cell. It has not been shown yet that children can inherit these types of genetic changes in tumor cells from their parents.
Symptoms
Similar to those of other tumours of the central nervous system (CNS), the presenting symptoms of ependymoma primarily depend on the patient’s age, tumour site and size as well as pattern of spread within the CNS. The following general (nonspecific) and local (specific) symptoms can occur:
General (nonspecific) symptoms
Unspecific general symptoms occur independently of the tumour’s location. They may be similar to and therefore mimic other, non-CNS diseases. General symptoms of a child or adolescent with a CNS tumour may include headaches and/or back pain, dizziness, loss of appetite, nausea and vomiting (particularly after getting up in the morning), weight loss, increasing fatigue, inability to concentrate, school problems, mood swings, and character changes as well as developmental delay, to name a few.
Major reason for these symptoms is the slowly but continuously increasing intracranial pressure (ICP). Elevated ICP may be caused by the growing, thus more and more space-occupying tumour within the bony skull, but also by the tumour blocking the regular flow of the cerebrospinal fluid, thereby forming hydrocephalus. In babies or small children with soft spots (open fontanelles), elevated intracranial pressure and hydrocephalus typically present with a bulging fontanelle or a larger than expected head circumference (macrocephalus), respectively.
Local (specific) symptoms
Local symptoms may indicate the tumour location and, thus, which functional regions of the CNS may be affected. Therefore, an ependymoma in the cerebellum can cause dizziness and gait disturbances, whereas such a tumour in the hemispheres of the cerebrum can be associated with seizures and/or motor deficits and a tumour of the spinal cord with different kinds of neurological impairments such as motor and sensory deficits as well as typical gait disturbances. Also, impaired vision, mental and sleep problems may, although to a lesser extent, be indicative of tumour location.
Good to know: Not all patients presenting with one or more of the symptoms mentioned above do have an ependymoma or another type of brain tumour. Many of these symptoms may also occur with other, harmless diseases that are not associated with a brain tumour at all. However, if certain symptoms persist or get worse (for example repetitive headaches or rapid increase of head circumference in a young child), a doctor should be seen to find the underlying reason. In case it is an ependymoma or some other brain tumour, treatment should be started as soon as possible.
Diagnosis
If the paediatrician thinks that the young patient’s history (anamnesis), physical examination and possibly even results from diagnostic imaging are suspicious of a tumour of the central nervous system (CNS), the child should immediately be referred to a hospital with a childhood cancer program (paediatric oncology unit), where further diagnostics can be initiated and performed by childhood cancer professionals. Very close collaboration between various specialists (such as paediatric oncologists, paediatric neurosurgeons, paediatric radiologists, to name a few) is required, both to find out, whether the patient really suffers from a malignant CNS tumour and, if so, to determine the tumour type and the extension of the disease. Knowing these details is absolutely essential for optimal treatment planning and prognosis.
Tests to secure diagnosis
The initial diagnostic procedures for a young patient presenting with a suspected CNS tumour at a childhood cancer centre include another assessment of the patient’s history, a thorough physical/neurological exam and imaging diagnostic, such as magnetic resonance imaging (MRI) or (less often) computed tomagraphy. These tests help to find out exactly whether the patient has a tumour of the central nervous system. Also, localisation and extent of the tumor, its demarcation regarding adjacent tissue as well as a potential hydrocephalus can be diagnosed by these imaging techniques very well.
In order to validate the final diagnosis, histological and molecular analysis of surgically obtained tumour tissue (biopsy) is required. Usually, this is done using the tissue obtained during surgical tumour removal.
The extent of histological and, especially, molecular genetic workup has been substantially increased over the past years. Today’s option of using modern laboratory techniques makes it possible to identify molecular tissue characteristics that do not only help finalize the diagnosis, but can also provide information on what to expect regarding the course of the disease (such as growth behaviour). Hence, molecular diagnostics already play an important role in treatment planning and will most certainly become even more relevant in the future.
Tests to assess spread of disease
Once the diagnosis of an ependymoma has been confirmed, additional tests are required to assess the extent of the disease within the central nervous system (CNS). Apart from MRI scans of the complete CNS (brain and spine) for macroscopic metastases, these tests also include microscopic checking of the cerebrospinal fluid (CSF) for tumour cells in the spinal cord (which are not visible by MRI scan). Cerebrospinal fluid is mostly obtained from the spine in the lower back (lumbar puncture), since the risk of the puncture needle damaging the spinal cord is lowest at the lower back level.
Tests before treatment begins
In preparation for the intensive treatment of the brain tumour, further investigations are performed, such as electrocardiography (ECG) and echocardiography to check cardio function. Furthermore, additional blood tests are needed to assess the patient’s general health condition and to check whether the function of certain organs (such as liver and kidneys) is affected by the disease and whether there are any metabolic disorders to be considered prior or during therapy. Any changes occurring during the course of treatment can be assessed and managed better based on the results of those initial tests, which thus help to keep the risk of certain treatment-related side effects as low as possible.
Also, the patient’s blood group needs to be determined in case a blood transfusion is required during treatment. In sexually mature females (which means after they have experienced their first menstrual bleeding), a pregnancy test is recommended prior to treatment as well.
Good to know: Not every patient needs the complete check-up. On the other hand, tests might be added that haven't been mentioned here, depending on the individual situation of the patient. Your caregivers will inform you and your child, which diagnostic procedures are individually required in your case and why.
Treatment planning
After the diagnosis has been confirmed, therapy is planned. In order to design a highly individual, risk-adapted treatment regimen for the patient, certain individual factors influencing the patient’s prognosis (called risk factors or prognostic factors) are being considered during treatment planning (risk-adapted treatment strategy). One important prognostic factor is the type (subtype) of ependymoma, which defines its grade of malignancy and is assessed by histological and molecular genetic analysis of a tumour sample. The currently used classification of ependymomas will be outlined later.
Further prognostic factors are the size, localization, and spread of the tumour, for these factors have an impact on whether or not the tumour can be completely removed by means of an operation, thus influencing the patient’s chances of survival. In addition, the patient’s age at the time point of diagnosis and his overall physical condition play an important role. All these factors are included in treatment planning in order to achieve the best possible outcome for each patient.
Classification of Ependymoma
There are various subtypes of ependymoma: they look different under the microscope, meaning histologically, and also present with different molecular characteristics.
Until recently, the classification of ependymomas was exclusively based on their histology. Classification according to the World Health Organization (WHO) histologically differentiated low-grade ependymomas (WHO grade I), which grow slowly, since their cells hardly ever divide, and higher-grade ependymomas (WHO grades II and III) with more frequently dividing cells and other typical characteristics of aggressive growth. However, the appearance of the tumours under the microscope has turned out not to be insufficient to predict their growth behaviour. Therefore, since 2016 classification has been based on molecular genetic (biological) tumour characteristics in addition to the histological features. This additional consideration of molecular factors has shown to provide a more precise assessment of the tumour biology and, thus, is more feasible for optimal treatment planning. Tthe molecular subtype of a tumour will, therefore, increasingly impact the choice of optimal treatment.
As per current classification of the World Health Organization (WHO classification) for tumours of the central nervous system, the following subtypes of ependymoma have been defined (regarding histological and molecular genetic criteria):
- Subependymoma WHO grade I: slowly growing, low-grade tumour
- Myxopapillary ependymoma grade I: slowly growing, low-grade tumour (mostly found in the spinal canal)
- Ependymoma WHO grade II: usually slowly growing tumour with only a few features of aggressive growth
- Ependymoma RELA fusion-positive WHO grade II oder III: tumour with partly aggressive growth behaviour
- Anaplastic ependymoma WHO grade III: tumour with aggressive behaviour.
Most ependymomas in children and adolescents are grade II- or grade III-tumours: WHO grade II-ependymoma as well as anaplastic ependymoma WHO grade III are dominating in the cerebellar region; in the hemispheres, ependymoma RELA fusion-positive WHO grade II and III are most common. The myxopappillary ependymoma WHO grade I is restricted to the spinal canal. Subependymomas WHO grade I are rare in childhood and adolescence.
Note: The cut-off between grade II- and grade III-tumours cannot always be clearly determined. Therefore, this classification cannot be used to precisely predict the growth behaviour of an individual tumour. However, there is no difference between WHO grade II- and III-ependymomas regarding treatment and prognosis.
Treatment
Treatment of children and adolescents with ependymoma should take place in a children's hospital with a paediatric oncology program. Only such a childhood cancer centre provides highly experienced and qualified staff (doctors, nurses and many more), since they are specialised and focussed on the diagnostics and treatment of children and teenagers with cancer according to the most advanced treatment concepts. The doctors in these centres collaborate closely with each other. Together, they treat their patients according to treatment plans (protocols) that are continuously optimised. The goal of the treatment is to achieve high cure rates while avoiding side effects as much as possible.
Current treatment concepts for children and teenagers with ependymoma include surgical tumour removal, radiotherapy and, for some patients, chemotherapy. Very young children receive chemotherapy prior to radiotherapy to delay the beginning of radiation.
Surgery
Surgical tumour removal plays an important role in the treatment of ependymoma, because the extent of tumour resection has a major impact on the subsequent course of the disease: complete tumour resection is usually associated with a more favourable prognosis than partial resection, which may be the only option in some patients with advanced disease, for example.
If complete resection was not achieved during primary surgery, the doctors will recommend a second attempt of resection as long as the risk of the procedure is justifiable. However, some ependymomas are located in parts of the brain that make complete resection impossible. In particular, tumours in the area of the fourth brain cavity (cerebral ventricle) and in the so-called cerebellopontine angle usually allow only partial removal. Complete resection would be associated with a high risk of damaging healthy, vitally important brain tissue.
Additional treatment (adjuvant therapy)
The decision-making regarding additional, non-surgical treatments is based, above all, on the histological tumour type (WHO grade) and the extent of surgical tumour removal. While a completely removed WHO grade I-ependymoma usually does not require additional therapy, patients with ependymomas WHO grade II or III located in the brain (intracranial ependymomas) are considered for adjuvant treatment, which usually includes radiation therapy of the tumour region. Some patients also receive chemotherapy (see below).
Chemotherapy uses drugs (so-called cytostatic agents or cytostatics) that can kill fast-dividing cells, such as cancer cells, or inhibit their growth, respectively. Radiotherapy is done using energy-rich, electromagnetic radiation, given through the skin to the tumour region. Radiation causes DNA damage in tumour cells, thereby leading to cell death. Aside from this so-called conventional radiotherapy, particle-radiation with protons (also known as proton therapy) can be an option for some patients as well. This type of radiotherapy provides the benefits of better targeting the tumour area, thus sparing more adjacent, healthy tissue from the effects of radiation. Proton therapy is gaining an increasing importance in the treatment of children and teenagers with solid tumour.
Therapy options for patients with ependymoma WHO grade I
Patients with a myxopapillary ependymoma grade I of the spinal cord, whose tumour could be completely removed, usually do not need additional treatments. However, they are regularly seen by their caregiver team for physical exams and imaging controls. In case there is remaining tumour after surgery, radiotherapy of the tumour region has proven to be of benefit and is given upon decision on an individual basis.
Other WHO grade I ependymomas are rare in children. Hence, decision-making regarding optimal treatment is based on the patient’s individual situation.
Therapy options for patients with ependymoma WHO grade II-III
For patients with ependymomas presenting with signs of malignancy such as ependymoma WHO grade II, anaplastic ependymoma WHO grade III or ependymoma RELA fusion-positive WHO grade II or III, respectively, additional treatments are indicated, even if complete tumour resection was achieved. The reason beyond this strategy is that, despite macroscopic (that means visible) total removal, remaining tumour cells, which are invisible to any surgeon’s eye, may be left in the body. These remainders are associated with a high risk of recurrent disease at the original tumour site (local relapse). Recurrent disease at other sites of the brain or spinal cord is rather rare.
Radiotherapy, and, for some patients, additional chemotherapy, help reduce the risk of local (recurrence). Chemotherapy is particularly given to very young children, in order to delay or even completely avoid radiotherapy while they are so young. Also, treatment intensification (for example in case of metastasis or remaining tumour) can be a goal of chemotherapy.
The doctors will recommend the type of additional treatment based on the type of ependymoma, its grade of spread, the extent of surgical removal and the patient’s age at diagnosis.
According to the current treatment recommendations of the HIT-MED study centre (HIT-MED-Guidance), patients 18 months old and older who neither have remaining tumour nor metastases after surgery usually receive radiotherapy of the tumour region. Younger patients without metastases or remaining tumour, respectively, receive an intensive chemotherapy, sometimes followed by radiation of the tumour region once they have turned one year old.
For patients with remaining tumour after initial surgery, the goal of non-surgical (adjuvant) treatment is to shrink the remaining tissue in order to fa-cilitate a second attempt of complete tumour removal. This can be achieved by an intensive chemotherapy. Depending on the outcome and the patient’s age, more cycles of chemotherapy as well as radiotherapy of the remaining tumour are further options. Patients with metastases (which are rare at primary diagnosis) receive individual treatment under the supervision of the HIT-MED study centre.
Note with regards to intraspinal ependymas WHO grades II and III: patients with ependymomas WHO grade II or III located in the spine may also receive radiotherapy (with or without chemotherapy). However, there are no standardized treatment recommendations for these patients and decision-making is based on the individual situation.
Note to the trial SIOP-Ependymoma II
Note to the trial SIOP-Ependymoma II: The therapy optimising study SIOP-Ependymoma II, which has been active in Germany since August 2019 (see next chapter on trials and registries), examines whether certain modifications of the previous standardized therapy result in improved outcomes. Hence, patients (after having provided informed consent) are randomized into different treatment arms, which enable the comparison between the current treatment recommendations and new options (standard arms and examining arms). Therefore, treatment of your child might differ from above-mentioned treatment concepts.
Therapy optimising trials and registries
In Germany, almost all children and adolescents with first diagnosis or relapse of ependymoma are treated within therapy optimising trials or registries. The term "therapy optimising trial" refers to a form of controlled clinical trial, which aims at improving current treatment concepts for patients based on the current scientific knowledge.
Patients who cannot participate in any study, for example because none is available or open for them at that time, or who do not meet the required inclusion criteria, respectively, may be included in a so-called registry. Such a registry pools scarce data in order to help with the planning of appropriate future clinical trials. To ensure optimal treatment for patients not registered in a study, experts from assigned trial panels usually provide recommendations and advice to the local caregiver team.
Currently, the following therapy optimising trials / registries are available for patients with ependymoma in Germany:
- SIOP-Ependymoma II: Since the beginning of 2019, children, adolescents and young adults with newly diagnosed ependymoma can be enrolled in the international, multi-centred trial SIOP-Ependymoma II. Approximately 60 paediatric oncology centres in Germany and numerous hospitals in other European countries are participating in this trial. The goal is to offer optimal and standardized diagnostics and treatment and to make research possible all over Europe.The German study centre is located at the Children’s Cancer Centre at the University of Hamburg. Principal investigator of the tral is Prof. Dr. med. Stefan Rutkowski.
- I-HIT-MED Registry: Patients with ependymoma, who for different reasons cannot or do not want to participate in any currently available or open trial, can be enrolled in this registry (International HIT-MED Registry), regardless of the treatment given. These patients will receive treatment as per individually designed treatment plans. The goal of the registry is not to assess the feasibility of an ongoing trial, safety or efficacy of a certain treatment. It rather aims at collecting individual patient data for future analysis. The headquarters of the registry are located in the Children’s Cancer Centre at the University of Hamburg, Germany (head of study: Prof. Dr. med. Stefan Rutkowski).
Prognosis
The prognosis of children and teenagers with ependymoma mainly depends on the tumour site and, thus, the extent of surgical tumour removal. Survival rates for patients after complete resection and subsequent radiotherapy are between 60 and 85 % after five years and between 50 and 70 % after 10 years, as long as the disease does not progress. After partial tumour removal, survival rates are far less favourable.
The overall prognosis for patients with intraspinal ependymoma is more favourable than for patients with ependymoma of the brain. However, remaining tumour as well as young age at diagnosis may have a negative impact on their prognosis.
When patients suffer from a recurrent ependymoma, opportunities for another surgery and/or radiation therapy will be evaluated. There is evidence that special irradiation techniques (such as stereotactic radiosurgery) can increase the average time of survival. Recurrent ependymomas are also sensitive to chemotherapy so that this form of treatment can improve the outcome for relapse patients.
Note: The survival rates mentioned in the text above are statistical values. Therefore, they only provide information on the total cohort of patients with these types of tumours. They do not predict individual outcomes.
In the context of cancer, the term „cure“ should rather be referred to as „free of cancer“, because current treatment regimens may help getting rid of the tumour, but they are also frequently associated with numerous late-effects. Early detection and appropriate management of these long-term sequelae typically requires intensive rehabilitation and thorough long-term follow-up care, although a patient may have been cured of the cancer.
References 
- Erdmann F, Kaatsch P, Grabow D, Spix C: German Childhood Cancer Registry - Annual Report 2019 (1980-2018). Institute of Medical Biostatistics, Epidemiology and Informatics (IMBEI) at the University Medical Center of the Johannes Gutenberg University Mainz 2020 [URI: https://www.kinderkrebsregister.de/ typo3temp/ secure_downloads/ 42507/ 0/ 1c5976c2ab8af5b6b388149df7182582a4cd6a39/ Buch_DKKR_Jahresbericht_2019_komplett.pdf]
- Fleischhack G, Rutkowski S, Pfister SM, Pietsch T, Tippelt S, Warmuth-Metz M, Bison B, van Velthoven-Wurster V, Messing-Jünger M, Kortmann RD, Timmermann B, Slavc I, Witt O, Gnekow A, Hernáiz Driever P, Kramm C, Benesch M, Frühwald MC, Hasselblatt M, Müller HL, Sörensen N, Kordes U, Calaminus G: ZNS-Tumoren. in: Niemeyer C, Eggert A (Hrsg.): Pädiatrische Hämatologie und Onkologie. Springer-Verlag GmbH Deutschland, 2. vollständig überarbeitete Auflage 2018, 359 [ISBN: 978-3-662-43685-1]
- vom Hoff, Katja: Therapieoptimierungsstudie SIOP-Ependymom II. WIR - Zeitschrift der Deutschen Kinderkrebsstiftung und der Deutschen Leukämie-Forschungshilfe e.V 2/17 [URI: https://www.kinderkrebsstiftung.de/ fileadmin/ Redaktion/ Zeitschrift_Wir/ 2017_2/ Therapieoptimirung.pdf]
- Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW: The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta neuropathologica 2016, 131: 803 [PMID: 27157931]
- Pajtler KW, Witt H, Sill M, Jones DT, Hovestadt V, Kratochwil F, Wani K, Tatevossian R, Punchihewa C, Johann P, Reimand J, Warnatz HJ, Ryzhova M, Mack S, Ramaswamy V, Capper D, Schweizer L, Sieber L, Wittmann A, Huang Z, van Sluis P, Volckmann R, Koster J, Versteeg R, Fults D, Toledano H, Avigad S, Hoffman LM, Donson AM, Foreman N, Hewer E, Zitterbart K, Gilbert M, Armstrong TS, Gupta N, Allen JC, Karajannis MA, Zagzag D, Hasselblatt M, Kulozik AE, Witt O, Collins VP, von Hoff K, Rutkowski S, Pietsch T, Bader G, Yaspo ML, von Deimling A, Lichter P, Taylor MD, Gilbertson R, Ellison DW, Aldape K, Korshunov A, Kool M, Pfister SM: Molecular Classification of Ependymal Tumors across All CNS Compartments, Histopathological Grades, and Age Groups. Cancer cell 2015, 27: 728 [PMID: 25965575]
- Frühwald MC, Rutkowski S: ZNS-Tumoren bei Kindern und Jugendlichen. Dtsch Arztebl Int 2011; 108: 390
- Merchant TE, Li C, Xiong X, Kun LE, Boop FA, Sanford RA: Conformal radiotherapy after surgery for paediatric ependymoma: a prospective study. The Lancet. Oncology 2009, 10: 258 [PMID: 19274783]
- Timmermann B, Kortmann RD, Kühl J, Rutkowski S, Dieckmann K, Meisner C, Bamberg M: Role of radiotherapy in anaplastic ependymoma in children under age of 3 years: results of the prospective German brain tumor trials HIT-SKK 87 and 92. Radiotherapy and oncology 2005, 77: 278 [PMID: 16300848]
- Timmermann B: Therapie von Ependymomen im Kindesalter - Eine aktuelle Übersicht. WIR Informationsschrift der Aktion für krebskranke Kinder e.V. (Bonn) 2002, 4: 21 [URI: http://www.kinderkrebsstiftung.de/ fileadmin/ KKS/ files/ zeitschriftWIR/ 2002_4/ behandlungsnetzwerk08.pdf]
- Timmermann B, Kortmann R, Kühl J, Meisner C, Bamberg M: Combined postoperative irradiation and chemotherapy for anaplastic ependymomas in childhood: results of the German prospective trials HIT 88/89 and HIT 91. Int J Radiat Oncol Biol Phys 2000, 46: 287 [PMID: 10661334]


 PDF Information on Ependymoma (476KB)
PDF Information on Ependymoma (476KB)